First-ever Clinical Study of Epigenetic Restoration
We received FDA authorization to proceed with first-in-human studies of ER-100 on January 15, 2026. This represents a landmark event for the field. Our Phase I study is designed to evaluate the safety and tolerability of ER-100 in optic neuropathies [open angle glaucoma (OAG) and non-arteritic anterior ischemic optic neuropathy (NAION)]. Because the study is in patients rather than healthy volunteers, we will also evaluate endpoints designed to explore efficacy using clinically meaningful measures of vision. For additional information about the clinical trial, please see clinicaltrials.gov.
Additional Indications
In addition to our lead program, we are exploring the potential of our epigenetic restoration platform in additional age-related indications.
We are developing appropriate delivery systems to enable OSK expression for these organ systems, demonstrating proof of concept in appropriate preclinical models, and identifying/advancing lead candidates.
Preclinical Proof-of-Concept (POC) in Metabolic Liver Disease
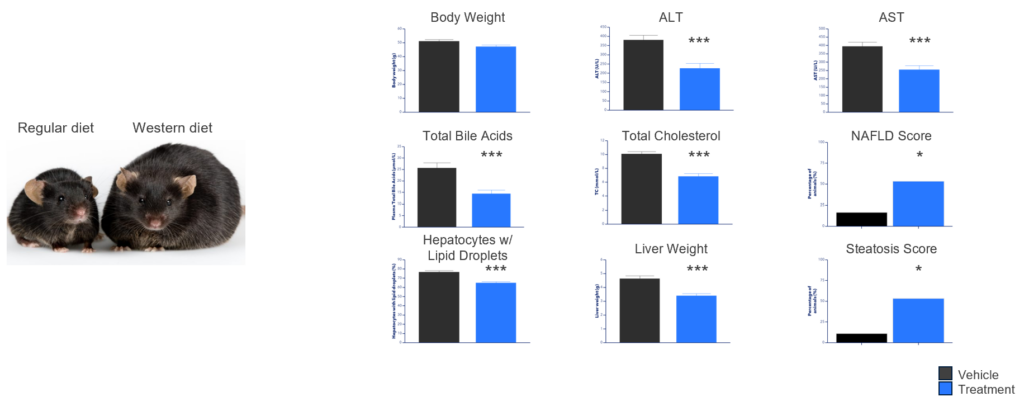
We have released data from preclinical studies of metabolic dysfunction-associated steatohepatitis (MASH) in a mouse model of diet-induced obesity (the Gubra GAN DIO-MASH model). MASH is a progressive, fibrotic liver disease and one of the most prevalent severe age-related diseases, with 10 million patients in the US alone.1
We achieved significant improvement in standard metabolic and clinically-derived biomarkers and histopathology in this preclinical model, evidence not only of therapeutic potential of epigenetic restoration in MASH, but also of the broad applicability of our platform.
MASH is a very different disease from OAG and NAION: while the eye is highly compartmentalized and neurons are post-mitotic, the liver has tremendous cross-talk with other organs and hepatocytes are some of the most rapidly dividing cells in the body. The fact that epigenetic restoration works preclinically in profoundly different organs and diseases is evidence of its potential as a broad platform.
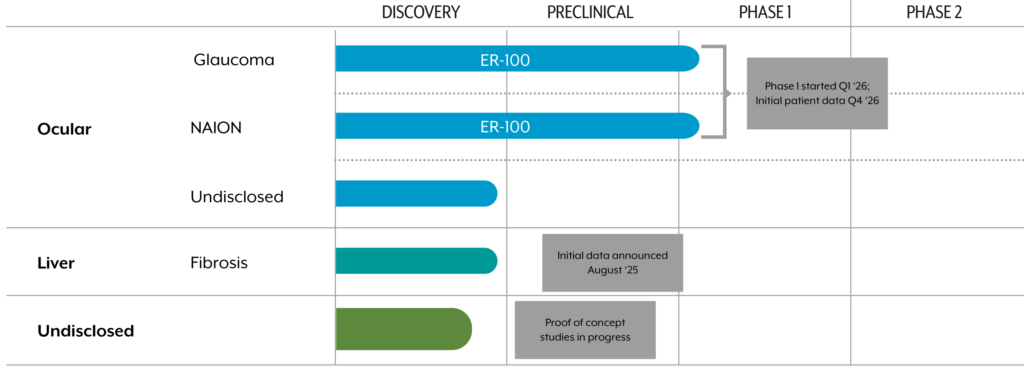
Overall Pipeline
Beyond treatments for OAG, NAION, and MASH, we are also advancing other candidates.