Optic Neuropathies
Life Biosciences’ lead candidate, ER-100 (AAV2-OSK), is in Phase I clinical trials for optic neuropathies. This is the first-ever partial epigenetic reprogramming candidate approved for clinical trials, and if successful, treatment with ER-100 would be the first time cells have been rejuvenated in humans.1,2
Optic neuropathies are a set of serious conditions that cause damage to the retinal ganglion cells (RGCs) of the eye, which are the neuronal cells responsible for transmitting visual signals to the brain. RGCs can be damaged by a range of age-related diseases, collectively termed optic neuropathies. Two such serious diseases are open-angle glaucoma (OAG) and non-arteritic anterior ischemic optic neuropathy (NAION).
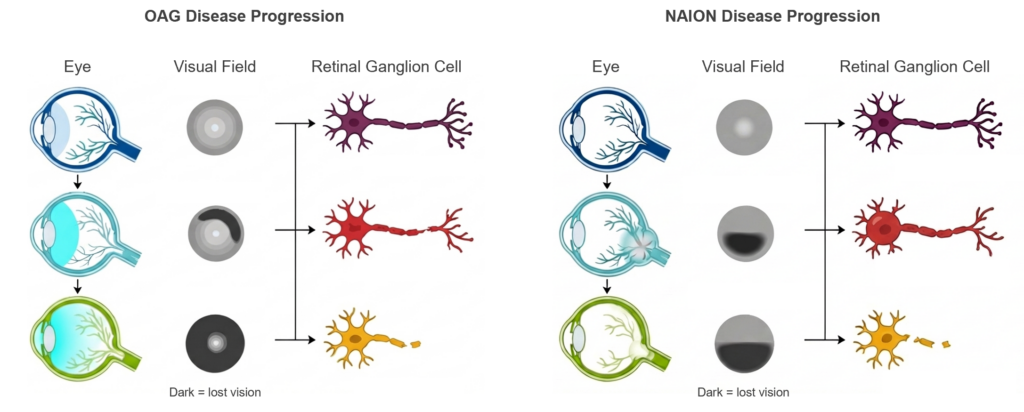
OAG involves damage to the optic nerve, often due to increased pressure inside the eye. Vision loss typically begins with subtle changes in peripheral (side) vision and progresses slowly over time. Because early stages often have no noticeable symptoms, many people are unaware they have OAG until significant vision loss has occurred. Even for those that catch OAG early, unfortunately, the disease can progress even with the best available treatments.
NAION is a sudden condition caused by reduced blood flow to the back of the eye. Unlike OAG, vision loss usually occurs quickly, often noticed upon waking, and typically affects one eye. The loss is often painless and may involve blurring or darkening in part of the visual field. Many people are left with permanent deficits, and there is a significant chance the other eye can be affected over time. Unfortunately, there are currently no approved treatments for NAION.
These diseases cause tremendous burden on patients, caregivers, healthcare systems, and society at large, both through the direct impact of vision impairment/blindness, and through the many consequences associated with vision impairment, including increased risk of institutionalization, higher incidence of falls, and increased social isolation leading to depression and dementia.
Addressing these diseases would bring enormous benefit to society… and especially to the patients who suffer from them.
2: Fatma Solmaz & Esra Tekin, “Inside the longevity boom”, Andolou Agency 2026
High Patient and Societal Burden of Optic Neuropathies

Extensive Preclinical Data on ER-100
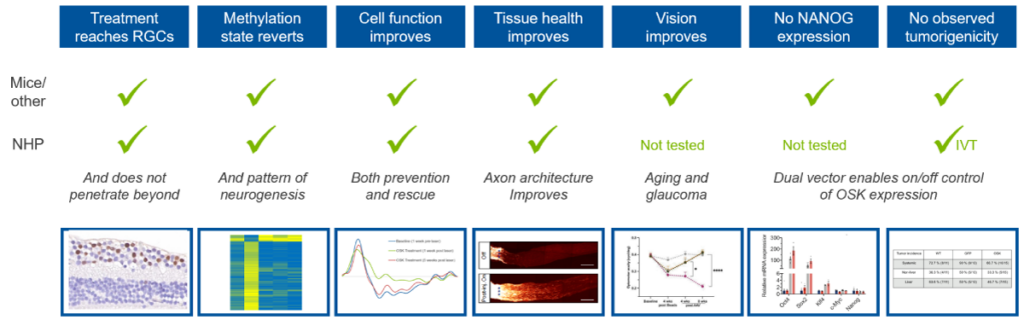
Life Biosciences has assembled a comprehensive set of data showing activity of ER-100 in preclinical models of optic neuropathies. Importantly, these data are not just from studies in mice and other rodents, but also in nonhuman primates (NHPs), which show much better likelihood of translation to human results.
Moreover, the data package covers a broad set of findings, all of which support the inference that ER-100 is working by partial epigenetic reprogramming to improve visual function.
As depicted in the graphic below, we have evidence in rodents and nonhuman primates that:
a) ER-100 enables expression of OSK in retinal ganglion cells (RGCs), which are the cells damaged in optic neuropathies
b) the epigenetic profile as measured by the DNA methylation state of genes changes with damage, but is restored with ER-100 treatment
c) ER-100 improves:
- visual function at the cellular level as measured by pattern electroretinogram (pERG)
- tissue health as measured by axon architecture
- vision as measured by optomotor response (OMR) (mice only)
Investigational new drug (IND)-enabling studies for biodistribution and toxicology were successfully completed and supported the advancement of ER-100 into human clinical trials. More information is available at clinicaltrials.gov.